Homemade dishwashing liquid soap. How I loathe thee...
Oh, but now ... I got you finally figured out!
Remember when I was not very impressed with how castile soap in my homemade dishwashing liquid soap was behaving? It left a cloudy film on my dishes and I was not too happy. You may have experienced it too. In fact, it did the same thing in my dishwasher soap formula so I had to come up with this revised formula. Sometimes, I had to rewash them and how un-eco-friendly (and frustrating) is it to rewash a load of pots and pans that you just finished washing???
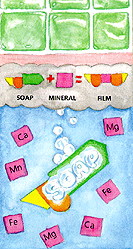
And do you know 'why' soap - especially castile soap - can leave a film? I explained in this post that soap reacts with mineral in the water and forms a film whereas detergents reacts less with mineral. And now, thanks to American Cleaning Institute (ACI), (Yes, there is such an institute in Washington D.C. It just might be my next favorite hangout!) you can understand exactly how it happens.
Although soap is a good cleaning agent, its effectiveness is reduced when used in hard water. Hardness in water is caused by the presence of mineral salts - mostly those of calcium (Ca) and magnesium (Mg), but sometimes also iron (Fe) and manganese (Mn). The mineral salts react with soap to form an insoluble precipitate known as soap film or scum.
But even though detergents are better for hard water, I wouldn't use it to wash dishes since it's not that healthy for us and for the environment.
So I used my own homemade dishwashing soap with washing soda (to soften the water and to thicken) in the past but lately, I was wondering if I should even use washing soda now.
I discovered that washing soda is caustic when I made my own a couple of weeks ago. Remember how I told you NOT to taste it because it's NOT edible? So I wondered if I should eliminate washing soda too just like how I eliminated Borax from the dishwasher "detergent" and the laundry detergent. Hmmm...
Well, I experimented and came up with a "new and improved" dishwashing liquid soap without borax and washing soda. It turns out washing soda IS used for water softener because it's a form of salt. So why not just use kosher salt? Right? Right. Less ingredients the better is my motto.
Borax Free Castile Soap Dishwashing Liquid Formula
Ingredients:
½ C Castile Soap
½ C Water
1 teaspoon Kosher Salt (to thicken and to soften water)
Equipment:
Jar or soap pump
Funnel
Pan
Spoon to stir
Instruction:
Combine the ingredients and gently heat in a pan while mixing. You'll see salt harden as soon as you add it to the pan. 

I had to put the bottle in hot water and melt it again. So if this happens to you; don't panic. Just warm the bottle in a bath of hot water and it will melt. Then, add a bit more water and you're good to go!
Here are some observations I made about using Castile soap for washing dishes.
Rinse - When washing the dishes with this soap, rinse quickly afterwards. Do not let a sink full of dishes sit in soapy water using castile soap. Longer it's in contact with castile soap, more it will react with the mineral, forming a cloudy film. That's the reason why sometimes using dishwashing detergent made with castile soap comes out filmy and cloudy. Work quickly to rinse.
Salt - salt is a water softener and it also creates suds. Washing soda is actually used as water softener, it turns out. And the reason I used it for the dishwasher is because I can add vinegar as a rinsing agent and it can help with mineral removal. But for hand washing, I don't like to use the washing soda since I don't have a chance to rinse with vinegar. Actually, that made me think of the next point...
Vinegar rinse - if you find the dishes to be cloudy, mix about ½ C of distilled white vinegar to about gallon of water. Soak the dishes for about 10 minutes and rinse them well. Using hot water can exacerbate the film so use cool water to rinse. If the dishes are still cloudy and you can't get the film off, use dishwashing liquid soap made by companies like Ecos or Ecover (plant derived and aquatic animal friendly) to wash them again. Once the film is stuck on the dishes, you have no choice but to use commercial dish soap to take them off. But with this formula, you shouldn't have problems as bad as using castile soap alone.
P.S. If you want to buy castile liquid soap (affiliate link) to try this recipe without waiting for the giveaway, there are many places you can buy, including your local supermarkets. But Amazon (affiliate link) and Drugstore (Not an affiliate) also carry them and are pretty inexpensive.
Drawing of the soap in water molecules used with permission from American Cleaning Institute








sarah says
Hi, I know this post is a little older, but I recently found this recipe, and it's the closest I've come to homemade dish soap I really like. I've got pretty hard water, so I am wondering if there is anything else I can add to this recipe to avoid water spots on my dishes (esp. glassware) if I let them air-dry. I've been looking all over the web, but I haven't had too much luck so far. I've tried adding additional salt to my washing water (I use the fill-the-sink method) and adding vegetable glycerin on a separate occasion, but not with the results I was hoping for. Any input would be fabulous!
Mrs Sarah says
Why not use Baking soda instead of Washing Soda or salt? Salt is Sodium Chloride, Washing Soda is Sodium carbonate, while baking soda is NOT caustic and is sodium bicarbonate (two parts in instead of one), and it is nearly 8 times cheaper in Bulk than salt is.
homeschoolmommyto3 says
How much of this would you use I. Your dishwasher?
MerryMessyLife says
Great post! So, just adding that 1 tsp. of kosher salt and heating it makes it thicken and work? I've tried using Castile Soap for handwashing detergent before and it also didn't work well for me. I learned a lot from your post. Thanks for sharing!
ecokaren says
MerryMessyLife Yes it does. Isn't it crazy? But apparently salt draws water out of the soap and since you are heating it, water evaporates. Just make sure to watch the amount of salt you are adding since it may end up thickening it too much, like the photo in the post. Let me know if this works for you. Thanks for reading!
Karmalades Household Cleaners with a Conscience says
Love this post!!
swiggett says
In homemade shampoo. Not my best recipe, but it works
amberpoppenga says
I have recipes for shampoo! Would love to win a bottle to try it out 🙂
lindaeve says
amberpoppenga I tried a Dr. Bronner's based shampoo and it just did not rinse out properly. Hard water problem I guess. Maybe with a vinegar rinse after?
ecokaren says
lindaeve if you have hard water, you should try the ACV rinse afterwards. I heard vinegar rinse makes your hair soft anyway...so if you use castile soap and you have hard water, it'd probably be best to use it.
lindaeve says
ecokaren lindaeve Karen, I will try that. I actually used ACV rinse this morning and it's amazing. No tangles and my hair feels really soft. That's after using Herban Lifestayle's shampoo bar, which I LOVE and swear by.
ecokaren says
amberpoppenga I'm about to post my recipe next week. It's a shampoo recipe for dry scalp and it works great.
lindaeve says
I use Dr. Bronners to make a gentle hand soap (mixed with water) and to make an all purpose surface cleaner (mixed with a lot of water and a little eucalyptus oil). The surface cleaner is really nice!
agodlyhomemaker says
For shampoo and soap.
ErinBartlett says
We use castile soap in a foaming hand soap dispenser for washing hands. Works great!
ecokaren says
ErinBartlett my kids love the almond castile soap for the foaming soap dispenser!
ajaquier1 says
Thanks for doing the leg work on this! I'll be giving it a try! I have replaced our normal shower soap with Bronner's Castile soap. Love this product!
kim rohwer says
I love this follow-up on the recipe...thank you!
kim rohwer says
I love this follow-up on the recipe...thank you!
queentob says
We use Castile soap to wash our car and bathe our dog!
ecokaren says
queentob Ha! never thought to use it on our car and the dog! i have to try for both! Thanks for the tip!
chandralm says
I have successfully used plain castille soap for almost everything. I guess I must not have hard water?
cdergaz says
Thanks for this Karen!! I have tried and tried with several recipes online using Dr. Bronners and find that my dishes always end up with a filmy residue (and my sink, yuk!) - or I end up with a clumpy mess...I was so frustrated and ended up reverting back to conventional (eco) dishwashing liquids. I am excited to try this version and hope it works! 🙂
ecokaren says
cdergaz the key is to dilute it with water but then thicken it with salt...if that makes sense...i think when you dilute it, it takes away the soap property a bit but you don't meed much to do the dishes, you know? Of course, if you have a super greasy pan, it's best to rinse it out as much as you can first, then, use castile soap to finish it off. Castile soap will never fully get rid of grease if you don't rinse out the grease first. Let me know if this works for you. And remember, don't soak anything with castile soap. It makes filmy residue worse.
MonicaPowell says
Use to wash your hair instead of shampoo
VbBg says
I honestly have not tried Castile soap. But my friend swears by it for bathing her baby with super sensitive skin.
ecokaren says
VbBg this is your chance to try it if you win! It's awesome! But even if you don't win, you should buy a small bottle and try it.
EvonCassier says
Combine this with your tutorial on how to make a mason jar pump dispenser and you'll never have to buy liquid dish soap again. Great info!
ecokaren says
EvonCassier he he....you know me. I like to cover all bases...:)